- by administrator
- 3 April 2018
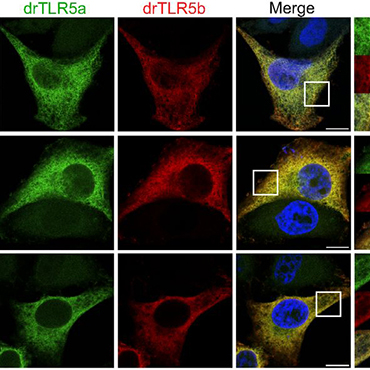
Duplicated TLR5 of zebrafish functions as a heterodimeric receptor.
Voogdt CGP, Wagenaar JA, van Putten JPM.
Proc Natl Acad Sci U S A. 2018 Apr 3;115(14):E3221-E3229.
Toll-like receptor 5 (TLR5) of mammals, birds, and reptiles detects bacterial flagellin and signals as a homodimeric complex. Structural studies using truncated TLR5b of zebrafish confirm the homodimeric TLR5-flagellin interaction. Here we provide evidence that zebrafish (Danio rerio) TLR5 unexpectedly signals as a heterodimer composed of the duplicated gene products drTLR5b and drTLR5a. Flagellin-induced signaling by the zebrafish TLR5 heterodimer increased in the presence of the TLR trafficking chaperone UNC93B1. Targeted exchange of drTLR5b and drTLR5a regions revealed that TLR5 activation needs a heterodimeric configuration of the receptor ectodomain and cytoplasmic domain, consistent with ligand-induced changes in receptor conformation. Structure-guided substitution of the presumed principal flagellin-binding site in human TLR5 with corresponding zebrafish TLR5 residues abrogated human TLR5 activation, indicating a species-specific TLR5-flagellin interaction. Our findings indicate that the duplicated TLR5 of zebrafish underwent subfunctionalization through concerted coevolution to form a unique heterodimeric flagellin receptor that operates fundamentally differently from TLR5 of other species.
